Steel metallurgy
Steel is the most widely utilized of the metallic alloy families. This ubiquity is owed to the varied microstructures of these alloys that can be tuned to achieve a uniquely broad range of properties. This versatility is most notably leveraged in sectors such as energy and transport. In these applications, the design of new steel grades and their processing is a hotter-than-ever topic where innovation is key in taking up modern society challenges. In the sector of energy for instance, oxide dispersion strengthened (ODS) steels bring a disruptive combination of strength and durability for fuel cladding in the next generation of nuclear reactors. In the same reactors, advanced dissimilar metal welds allow to join optimized alloys of different compositions to raise the safety and performance levels. In the sector of transport, transformation induced plasticity (TRIP) steels are a prime example of breakthrough in microstructure design that leads to considerable weight savings in automotive applications.
Those properties are controlled by the microstructures of the alloys, which intricately result from the combination of their chemical compositions and metallurgical processing. Our focus is on understanding the processing/microstructure/properties relationship in a quantitative way by combining advanced modelling and characterization techniques, such as in situ synchrotron X-ray diffraction or automated crystallographic orientation mapping (ACOM) in the TEM. Our research group is also committed to developing innovative methods to accelerate and optimize the design of new alloys. Combinatorial metallurgy is such a method where compositionally graded material fabrication and synchrotron facilities are leveraged to yield high throughput measurements of phase transformation kinetics.
(Alexis Deschamps, Frédéric de Geuser, Catherine Tassin, Hugo Van Landeghem, Muriel Veron)
Selected publications:
Those properties are controlled by the microstructures of the alloys, which intricately result from the combination of their chemical compositions and metallurgical processing. Our focus is on understanding the processing/microstructure/properties relationship in a quantitative way by combining advanced modelling and characterization techniques, such as in situ synchrotron X-ray diffraction or automated crystallographic orientation mapping (ACOM) in the TEM. Our research group is also committed to developing innovative methods to accelerate and optimize the design of new alloys. Combinatorial metallurgy is such a method where compositionally graded material fabrication and synchrotron facilities are leveraged to yield high throughput measurements of phase transformation kinetics.
(Alexis Deschamps, Frédéric de Geuser, Catherine Tassin, Hugo Van Landeghem, Muriel Veron)

Selected publications:
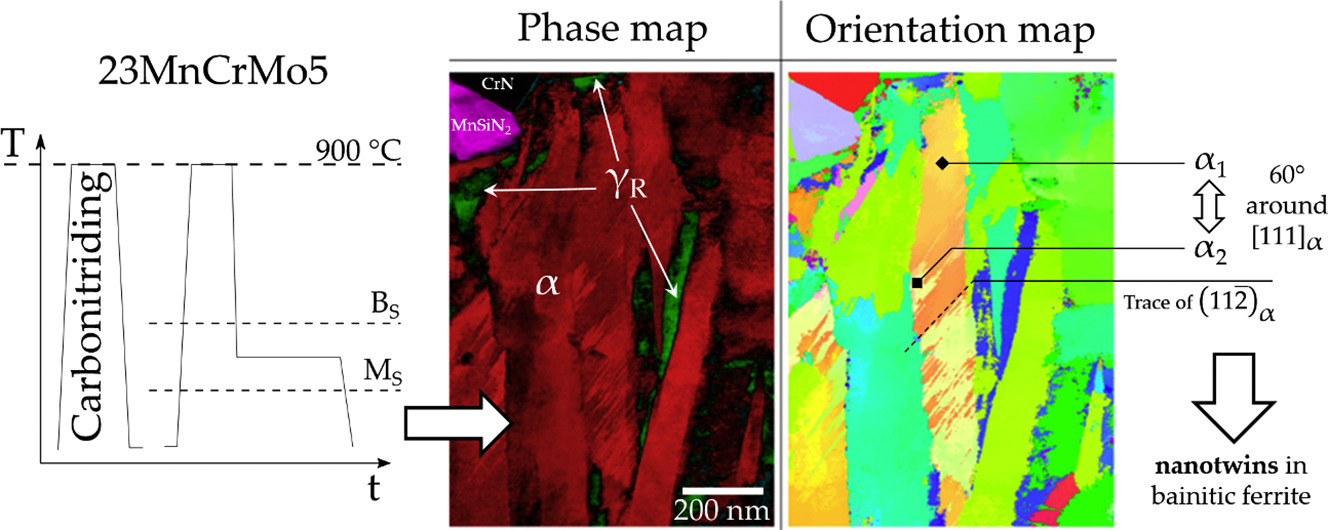
- Hugo P. Van Landeghem, Muriel Véron, Simon Catteau, Julien Teixeira, Jacky Dulcy, et al.. Nitrogen-induced nanotwinning of bainitic ferrite in low-alloy steel. Scripta Materialia, vol 155, pp. 63 (2018);
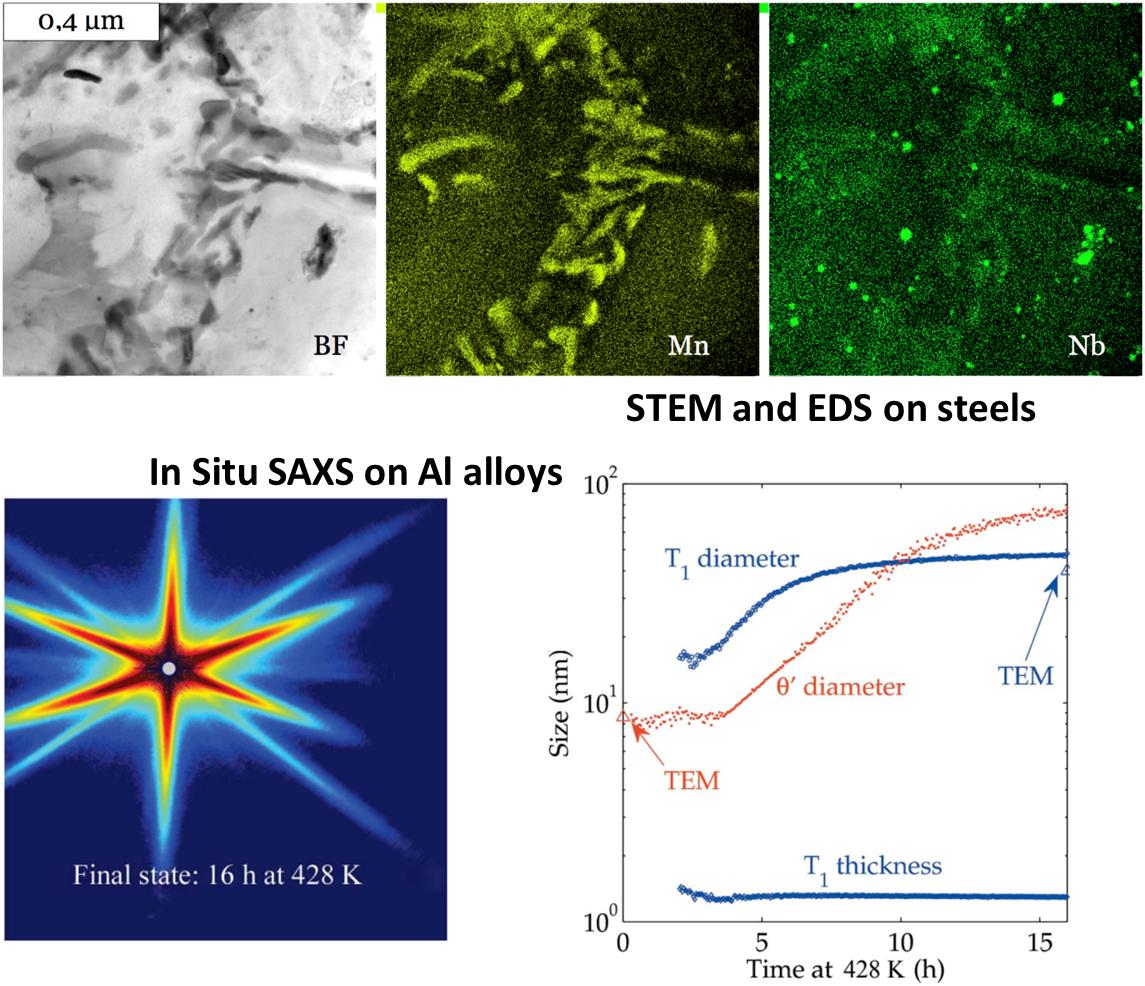
- Hasan Naser, Alexis Deschamps, Marc Mantel, Muriel Véron. Architectured duplex stainless steels micro-composite: Elaboration and microstructure characterization. Materials and Design, vol 145, pp. 156 (2018);
- A. Akhatova, F. Robaut, M. Verdier, M. Yescas, F. Roch, C. Tassin, H.P. Van Landeghem, Microstructural and mechanical investigation of the near fusion boundary region in thermally aged 18MND5 / alloy 52 narrow-gap dissimilar metal weld, Materials Science and Engineering: A, vol 788, pp 139592 (2020).
Precipitation in metallic alloys
Precipitation strengthening is one of the possible tools for optimising the mechanical properties of an alloy. We study precipitation from the earliest stages of decomposition of the solid solution to the formation of well-defined precipitates. We have a strong focus on aluminium alloys, particularly in relation with transportation industry (automobile, aerospace,…), but this topic is also oriented towards steels and other families of alloys (magnesium, titanium, nickel,...).
In addition to global experimental techniques such as mechanical testing or differential scanning calorimetry (DSC), this topic involves characterisation at the nanometre scale, which we achieve through transmission electron microscopy, as well as diffraction based techniques and small-angle scattering of X-ray (SAXS) both in the lab and at synchrotron sources and of neutron (SANS). SAXS experiments enables in particular in situ experiments and scanning of heterogeneous samples. A lot of the SAXS experiments is performed on the BM02-D2AM CRG beamline at ESRF (Grenoble), with which we have an historical institutional bond.
To better understand the phenomena at play, we used modelling tools at different scales. We study the thermodynamics of solid solutions and clustering in order to better understand nucleation in real alloys, we use meso-scale models to simulate precipitation kinetics and we study the dislocation-precipitates interactions to model their influence on the mechanical properties.
In addition to global experimental techniques such as mechanical testing or differential scanning calorimetry (DSC), this topic involves characterisation at the nanometre scale, which we achieve through transmission electron microscopy, as well as diffraction based techniques and small-angle scattering of X-ray (SAXS) both in the lab and at synchrotron sources and of neutron (SANS). SAXS experiments enables in particular in situ experiments and scanning of heterogeneous samples. A lot of the SAXS experiments is performed on the BM02-D2AM CRG beamline at ESRF (Grenoble), with which we have an historical institutional bond.
To better understand the phenomena at play, we used modelling tools at different scales. We study the thermodynamics of solid solutions and clustering in order to better understand nucleation in real alloys, we use meso-scale models to simulate precipitation kinetics and we study the dislocation-precipitates interactions to model their influence on the mechanical properties.
(Frédéric De Geuser, Alexis Deschamps, Patricia Donnadieu, Joel Lepinoux)
Selected publications:
- Laurent Couturier, Frédéric De Geuser, Alexis Deschamps Microstructural evolution during long time aging of 15–5PH stainless steel Materialia, vol 9, pp. 100634 (2020);
- Eva Gumbmann, Frédéric De Geuser, Christophe Sigli, Alexis Deschamps Influence of Mg, Ag and Zn minor solute additions on the precipitation kinetics and strengthening of an Al-Cu-Li alloy Acta Materialia, vol 133, pp. 172 (2017);
- Joel Lepinoux, Christophe Sigli Multiscale modelling of precipitation in concentrated alloys: from atomistic Monte Carlo simulations to cluster dynamics I thermodynamics Philosophical Magazine, vol 98, pp. 1 (2018).
High-temperature oxidation
This axis aims at bridging high temperature oxidation behavior of alloys with their composition, microstructure and thermo-mechanical history.
The formation of a protective oxide scale depends on the alloys composition as well as their structural and microstructural features which influence the diffusion of scale forming elements. The adhesion properties of the oxide scales are also crucial for long-term oxidation resistance.
We study the mechanisms giving rise to breakaway oxidation of stainless steels as the results of the fast growth of non-protective iron-rich nodules. Parameters of interest are: alloys composition and microstructure, composition of the oxidizing atmosphere, oxidation duration and temperature.
Adhesions properties of oxide scale are evaluated using micro-mechanical tests. Experimental results can be associated with finite element simulations based on micromechanical modeling of the oxidation affected zone.
(Valerie Parry, Celine Pascal, Muriel Braccini)
More in detail: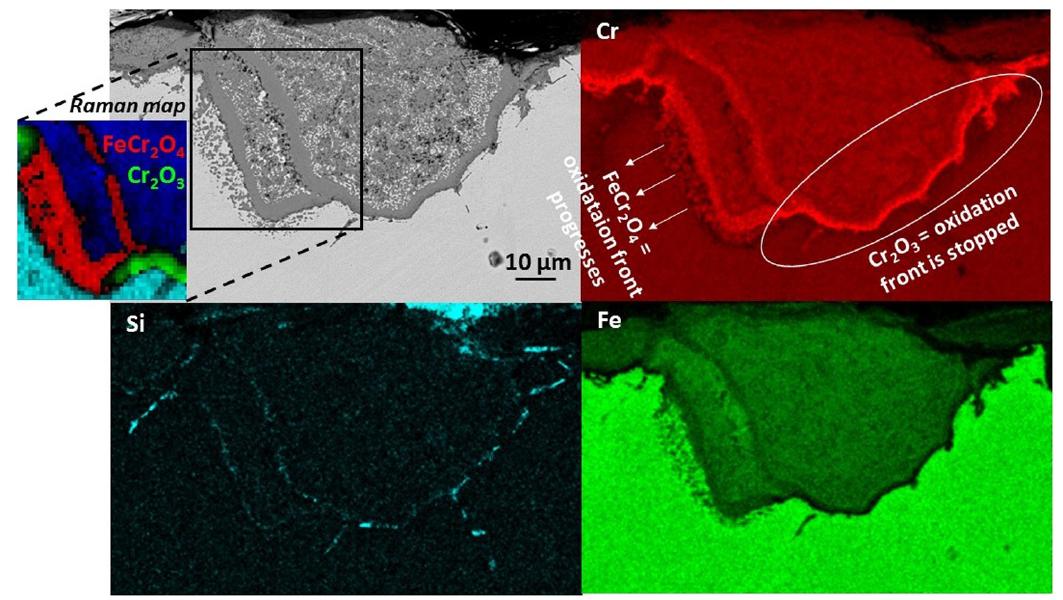
Selected publications:
The formation of a protective oxide scale depends on the alloys composition as well as their structural and microstructural features which influence the diffusion of scale forming elements. The adhesion properties of the oxide scales are also crucial for long-term oxidation resistance.
We study the mechanisms giving rise to breakaway oxidation of stainless steels as the results of the fast growth of non-protective iron-rich nodules. Parameters of interest are: alloys composition and microstructure, composition of the oxidizing atmosphere, oxidation duration and temperature.
Adhesions properties of oxide scale are evaluated using micro-mechanical tests. Experimental results can be associated with finite element simulations based on micromechanical modeling of the oxidation affected zone.
(Valerie Parry, Celine Pascal, Muriel Braccini)
More in detail:
- Austenitic stainless steels are known to be more susceptible to breakaway oxidation than ferritic one due to the lower Cr diffusivity in fcc-austenite than in bcc. The combination of compositional/elemental (TEM,Raman spectroscopy) and structural (EBSD) mapping techniques allowed investigating microstructural evolution of the oxide scale grown on Fe-17.5Cr-8.1Ni (in wt.%) oxidized at 850 C in O2 flow. The formation of banded inward-grown microstructure with alternated Cr-rich dense oxide scale (FeCr2O4 or Cr2O3 depending on the Cr content in the surrounding alloy) and internal oxidation zones (Fe–Cr spinel oxide and Ni–Fe metallic phase) was linked with the gradual Fe enrichment of the protective Cr2O3 chromia dense layer and its conversion in less protective FeCr2O4 spinel (See image below: SEM-BSE cross-section micrographs, SEM-EDX element mapping and Raman spectral mapping after oxidation of Fe-17.5Cr-8.1Ni at 850 °C in O2 flow for 13 days).

- Grain refinement via recrystallization process is a route for improving high temperature oxidation resistance of austenitic stainless steels. The effect of a pre-straining at room temperature on high temperature resistance of Fe-17.5Cr-8.1Ni (in wt.%), a low stacking fault energy austenitic stainless steel, was investigated at 830 C in O2 flow for 12 days. The formation of numerous grains supplying a network of Cr fast diffusion paths and a high density of oxide nucleation sites resulted in a dense fine-grained oxide scale which has limited the transport of scale-forming elements and moderated the long-term Cr consumption giving rise to a better oxidation resistance of the pre-strained substrate (See image below: beneficial effect of cold-working on high temperature oxidation resistance of Fe–17.5Cr–8.1Ni oxidized at 830°C for 13 days).
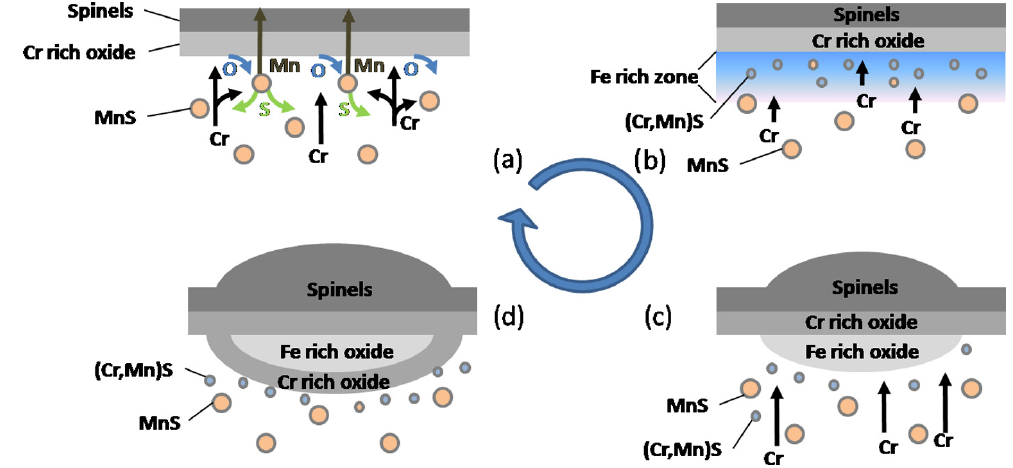
- Addition of sulphur improves steels machinability but decreases their resistance to high temperature oxidation. Experiments performed on Fe-17.1Cr-8.2Ni-0.3S steel (in wt.%) at 1000 C in O2 for 50 h associated with thermodynamic calculations point out a breakaway oxidation worsen by alloyed suphur. Thermal processing induce the partial dissociation of MnS inclusions and the formation of Cr,Mn-oxysulphides aggregates leading to a decrease of available Cr content in the bulk, below the oxide scale, needed to form the Cr-rich protective scale (See image below: schematic of breakaway oxidation induced by alloyed sulphur).
- Most industrial heat-resistant stainless steels contain silicon as a minor constituent. At high temperature, the internal formation of amorphous silica reduces oxidation rates but decreases the metal/oxide interface toughness. Tensile testing experiments performed on Fe-19Cr-9Ni steel (in wt.%) previously oxidized at high temperature showed a relation between the silica morphology and location and the damaging pattern. Calculations based on a micromechanical modeling using cohesive zone models to describe interfaces fracture behavior have shown that location of silica inclusions and silica/metal interface toughness are key parameters determining the cracks pattern morphology and the critical strain at failure (See image below: relations between oxidation induced microstructureand damaging pattern after tensile testing of Fe–18Cr–9Ni steel previously oxidized).
Selected publications:
- Valérie Parry, Audrey Col, Céline Pascal Beneficial effect of cold-working on high temperature oxidation resistance of austenitic stainless steel Corrosion Science, vol 160, pp. 108149 (2019);
- Audrey Col, Valérie Parry, Céline Pascal Oxidation of a Fe–18Cr–8Ni austenitic stainless steel at 850°C in O2: Microstructure evolution during breakaway oxidation Corrosion Science, vol 114, pp 17 (2017);
- Valérie Parry, Céline Pascal, Muriel Braccini et al. Relations between oxidation-induced microstructure and mechnanical durability Oxidation of Metals, vol 88, pp. 29 (2017).